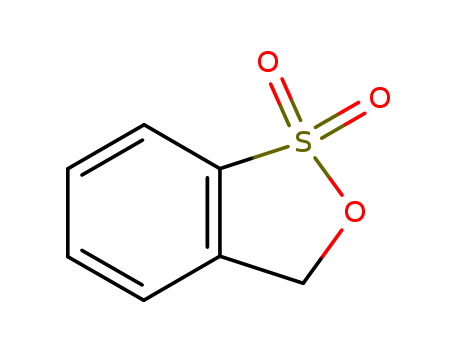
3H-2,1-benzoxathiole 1,1-dioxide
- CasNo:4430-23-3
Product Description
Chinese Manufacturer Supply 3H-2,1-benzoxathiole 1,1-dioxide 4430-23-3 On Stock with Competitive Price
- Molecular Formula:C7H6O3S
- Molecular Weight:170.189
- Vapor Pressure:0.00108mmHg at 25°C
- Refractive Index:1.602
- Boiling Point:310.7 °C at 760 mmHg
- Flash Point:141.7 °C
- PSA:51.75000
- Density:1.465 g/cm3
- LogP:1.98630
3H-2,1-benzoxathiole 1,1-dioxide(Cas 4430-23-3) Usage
|
General Description |
3H-2,1-benzoxathiole 1,1-dioxide, also known as dibenzothiophene dioxide, is a chemical compound with the molecular formula C12H8O2S. It is a heterocyclic organic compound containing both sulfur and oxygen atoms in a six-membered ring structure. 3H-2,1-benzoxathiole 1,1-dioxide is commonly used as a precursor in the synthesis of pharmaceuticals and agrochemicals. It is also used as a building block in the production of dyes and pigments. Additionally, dibenzothiophene dioxide has applications in the field of material science, where it is utilized in the synthesis of polymers and resin composites. Overall, this chemical compound has a wide range of industrial and scientific applications due to its unique molecular structure and reactivity. |
InChI:InChI=1/C7H6O3S/c8-11(9)7-4-2-1-3-6(7)5-10-11/h1-4H,5H2
4430-23-3 Relevant articles
Mixed carboxylic-sulfonic anhydride in reaction with imines: A straightforward route to water-soluble β-lactams via a Staudinger-type reaction
Bakulina, Olga,Dar'In, Dmitry,Krasavin, Mikhail
, p. 3989 - 3998 (2018/06/08)
The first example of employing a mixed c...
OXIDATION OF METHYL-SUBSTITUTED BENZENESULFONYL FLUORIDES IN THE PbO2-HSO3F SYSTEM
Arapov, O. V.,Rudenko, A. P.,Zarubin, M. Ya.
, p. 152 - 163 (2007/10/02)
The transformations of the sulfonyl fluo...
4430-23-3 Process route
-
-
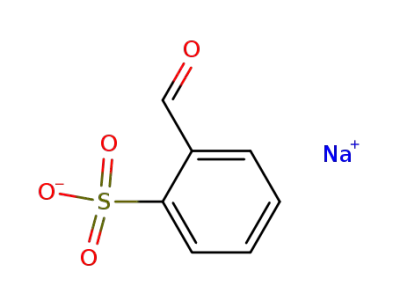
1008-72-6
sodium 2-formylbenzenesulfonate

-
![3H-benzo[c][1,2]oxathiole-1,1-dioxide](/upload/2026/4/22b51368736c41b5bf9ee169c6c6799a_9ab5271b-998f-4eae-a21a-cad2f6e51a1f.png)
-
4430-23-3
3H-benzo[c][1,2]oxathiole-1,1-dioxide
| Conditions | Yield |
|---|---|
|
Multi-step reaction with 2 steps
1: trichlorophosphate; phosphorus pentachloride / 1 h / 120 °C
2: hydrogenchloride; zinc / diethyl ether; water / 0.5 h / Heating
With
hydrogenchloride; phosphorus pentachloride; zinc; trichlorophosphate;
In
diethyl ether; water;
|
-
-
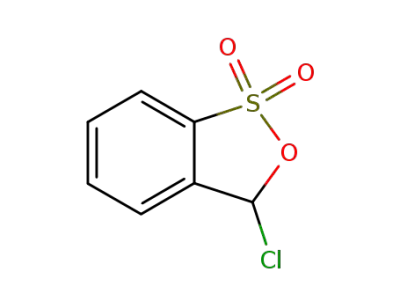
25595-59-9
3-chloro benzisoxathiole-1,1-dioxide

-
![3H-benzo[c][1,2]oxathiole-1,1-dioxide](/upload/2026/4/22b51368736c41b5bf9ee169c6c6799a_9ab5271b-998f-4eae-a21a-cad2f6e51a1f.png)
-
4430-23-3
3H-benzo[c][1,2]oxathiole-1,1-dioxide
| Conditions | Yield |
|---|---|
|
With
hydrogenchloride; zinc;
In
diethyl ether; water;
for 0.5h;
Heating;
|
13.4 g |
4430-23-3 Upstream products
-
444-31-5
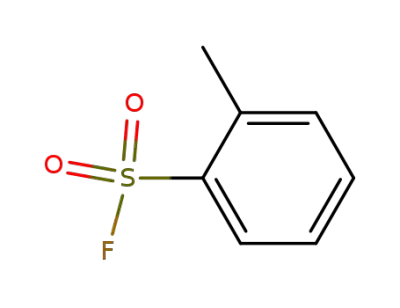
2-methylbenzene-1-sulfonyl fluoride
-
7647-01-0
hydrogenchloride
-
60-29-7
diethyl ether
-
62574-72-5
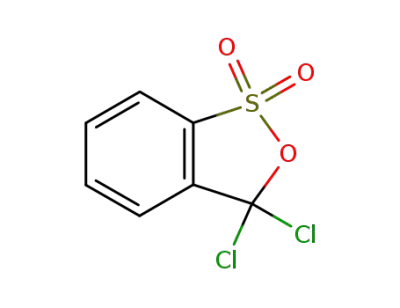
3,3-dichloro-3H-benz[c][1,2]oxathiol 1,1-dioxide
4430-23-3 Downstream products
-
3340-21-4
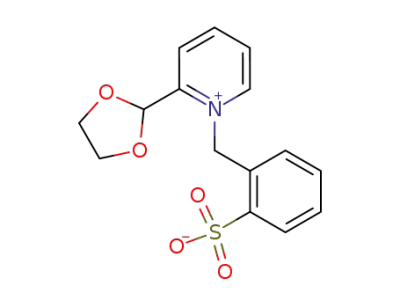
1-(2-Sulfo-benzyl)-2-<1,3-dioxolanyl-(2)>-pyridiniumhydroxyd-betain
-
3340-27-0
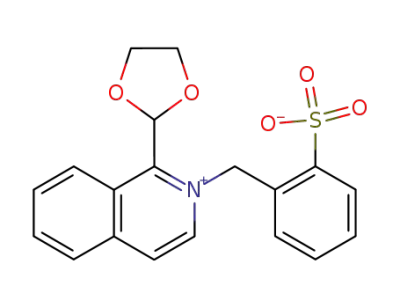
1-[1,3]dioxolan-2-yl-2-(2-sulfo-benzyl)-isoquinolinium betaine
-
3340-19-0
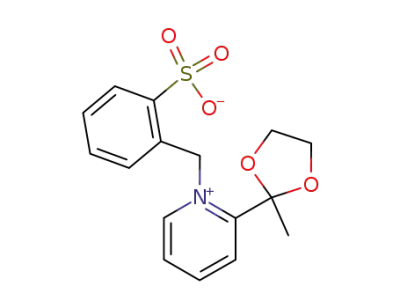
1-(2-Sulfo-benzyl)-2-<2-methyl-1,3-dioxolanyl-(2)>-pyridiniumhydroxid-betain
-
127324-81-6
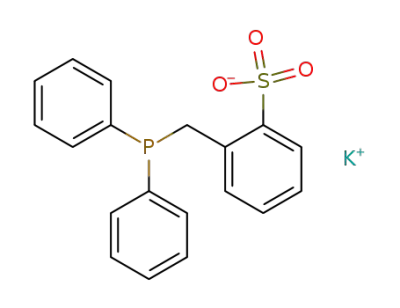
Diphenyl-(2-kaliumsulfonatobenzyl)-phosphin